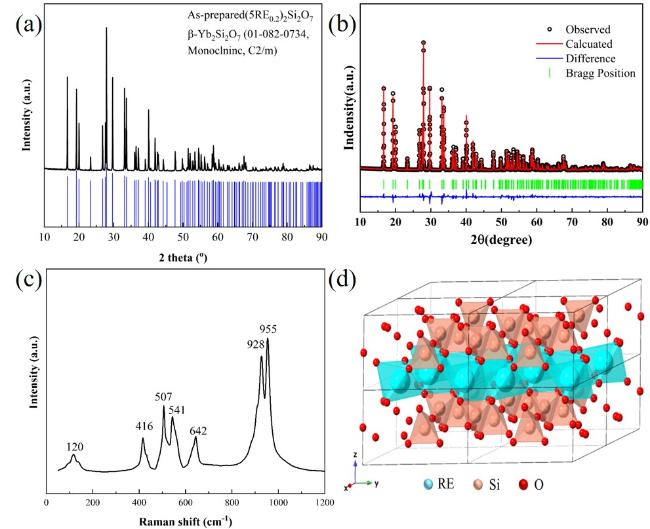
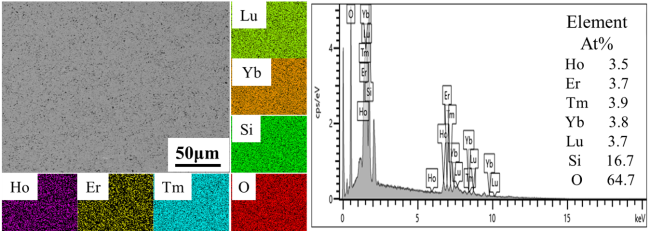
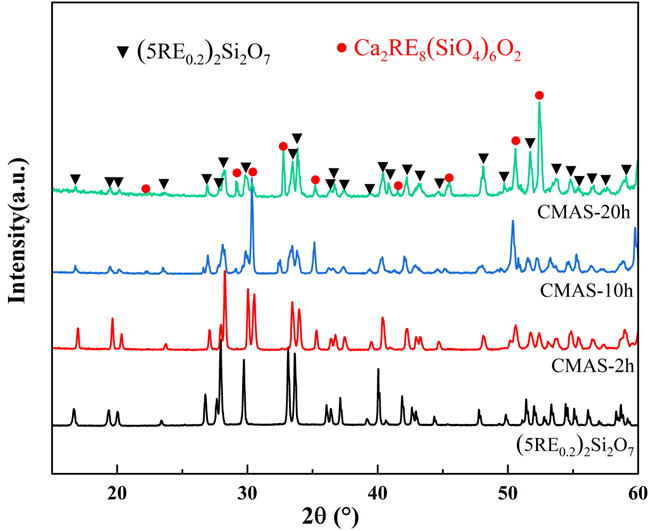
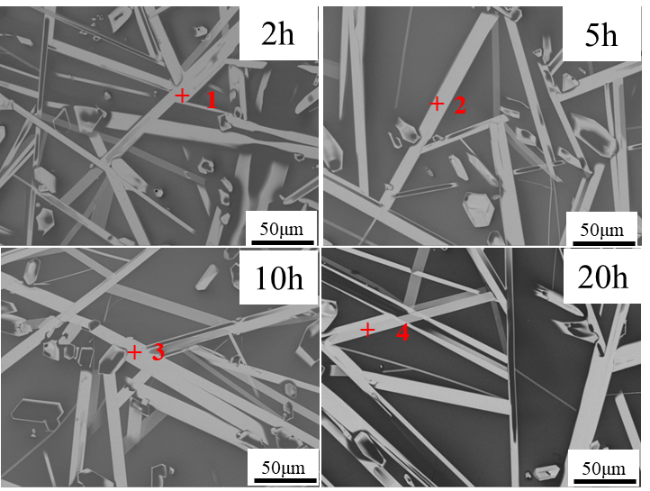
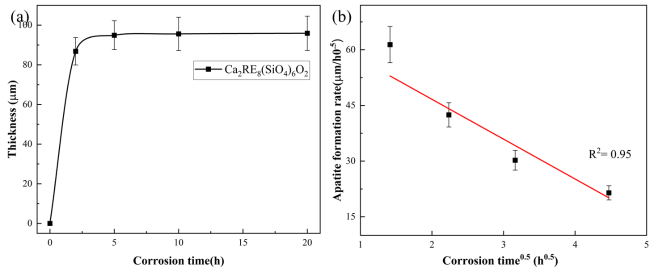
The increasing operating temperatures of high-performance engines have received significant interest in the thermal insulation properties of high-entropy ceramics for their applications in thermal/environmental barrier coatings
[21,24 -25]. High thermal insulation, characterized by low thermal conductivity, is crucial for reducing the operating temperature of the hot-end components and extending their service life. The specific heat capacity (
Cp) of the high-entropy (5RE
0.2)
2Si
2O
7 was calculated based on RE
2O
3 and SiO
2 constituent components using the Neumann-Kopp rule.
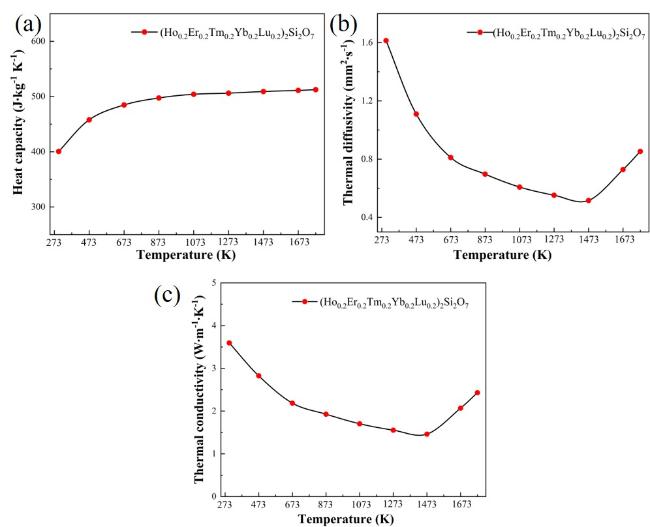
Fig. 4(a) shows the
Cp of high-entropy (5RE
0.2)
2Si
2O
7 from room temperature to 1500 ℃. The
Cp increases with the increase in temperature and gradually stabilizes above 800 ℃. The thermal diffusivity coefficient (α) of (5RE
0.2)
2Si
2O
7 ceramic decreased with increasing temperature from room temperature to 1200 °C and then increased with increasing temperature, as seen in
Fig. 4(b). The bulk density (ρ) of high-entropy (5RE
0.2)
2Si
2O
7 bulk is 5.5609 g/cm
3 based on the Archimedes drainage method. The thermal conductivity (λ
0) of high-entropy (5RE
0.2)
2Si
2O
7, as shown in
Fig. 4(c), was calculated using Eq. (2-3). The λ
0 has the same trend as
Cp with temperature changes. Phonon harmonic scattering and thermal radiation are the primary mechanisms influencing thermal conductivity. Phonon harmonic scattering is the dominant factor affecting the thermal diffusion coefficient and thermal conductivity below 1200 °C
[20]. Increased phonon collisions with rising temperature enhance phonon scattering, resulting in a significant decrease in the mean free path of the phonons, and consequently decrease α and λ
0 [24,26]. On the other hand, the influence of thermal radiation dominates above 1200 °C, resulting in an increase in both α and λ with increasing temperature. The thermal conductivity of high-entropy (5RE
0.2)
2Si
2O
7 ceramic increased from 1.46 to 2.43 W·m
−1·K
−1 with increasing temperature from 1200 °C to 1500 °C
[21,23].