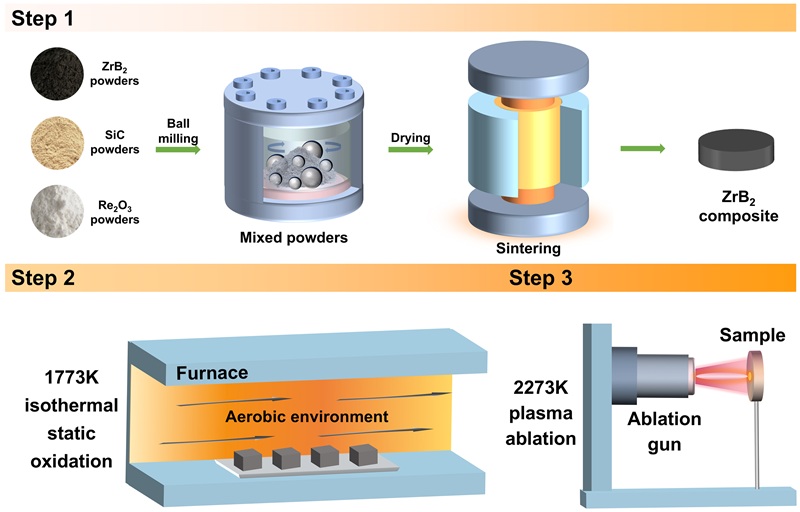
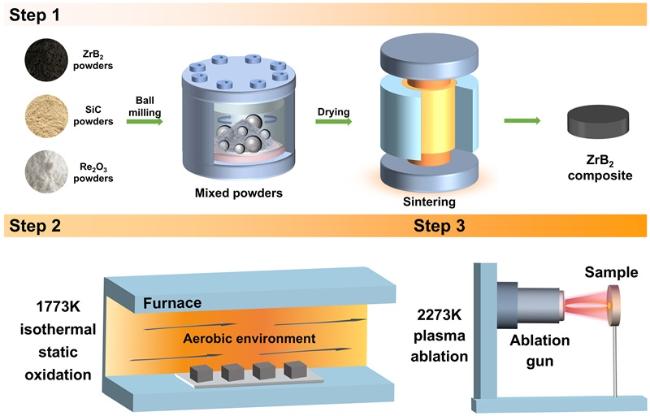
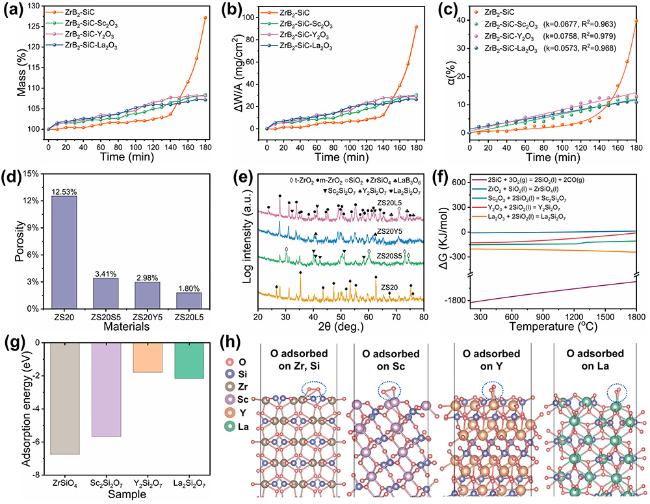
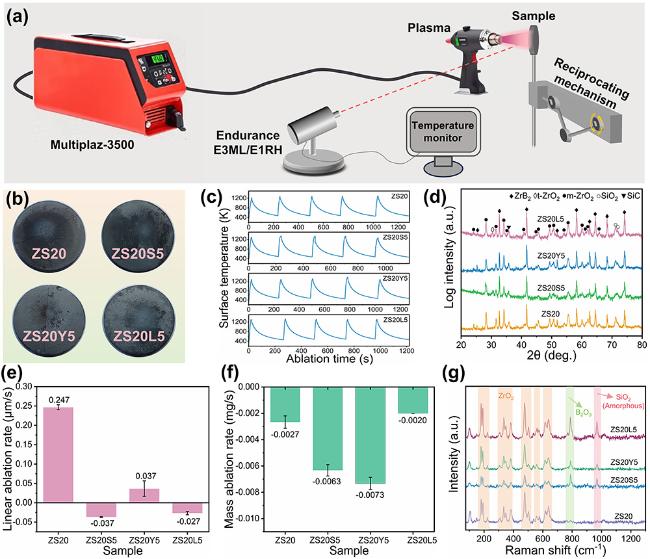
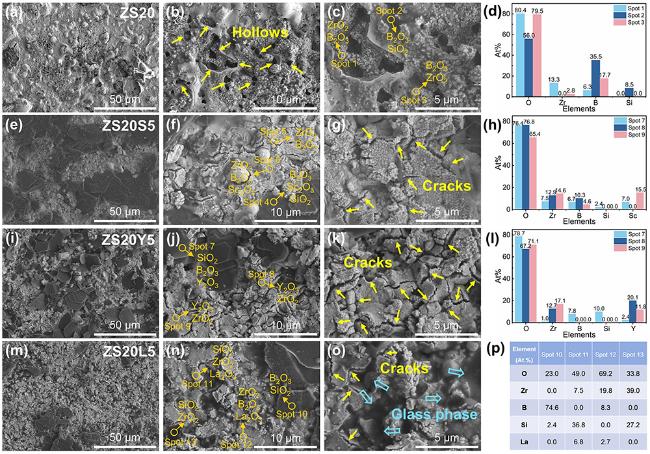
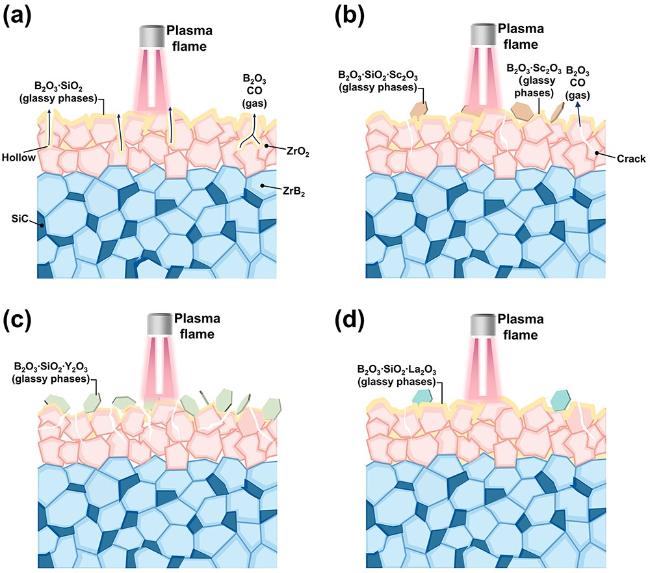
The surface microstructure and elemental distribution of oxidized ZS20 are shown in
Fig. 5a-d. After oxidation, ZS20 was converted into ZrO
2, B
2O
3, and SiO
2, which subsequently reacted at high temperatures to form a protective layer composed of ZrSiO
4 and borosilicate glass phases. The oxidized surface of ZS20 is distributed inhomogeneous protuberances and holes, which may be attributed to the volume expansion of oxides and the escape of the low melting point B
2O
3 and CO gases [
45,
46]. The oxidized surface of ZS20S5 is relatively flat compared with ZS20 (
Fig. 5e-g), and the results of EDS mapping and point scanning reveal that the primary component of the glassy phase is SiO
2. The granular phases correspond to ZrO
2, while the surface is coated with B
2O
3. Sc
3+ is mainly distributed within the ZrO
2 aggregates, and a portion of Sc
3+ may have dissolved into the ZrO
2 lattice at high temperatures, which induces the formation of t-ZrO
2. This can also be explained by the fact that the oxidized ZS20S5 sample contains a higher content of t-ZrO
2, and the content of t-ZrO
2 is regulated by the solid solution of rare earth elements in the ZrO
2 lattice [
47,
48]. The volumetric changes during the phase transition result in the formation of protrusions. Furthermore, Sc
2O
3 can react with SiO
2 to form Sc
2Si
2O
7 at high temperatures, and the distribution of Sc
2Si
2O
7 in B
2O
3 can effectively increase the glass viscosity, thereby reducing the B
2O
3 volatilization [
49]. As presented in
Fig. 5h-j, the oxidized surface of ZS20Y5 exhibits a uniform distribution of ZrO
2 particles, accompanied by discontinuous borosilicate glass phases, with no obvious glassy protective layer observed. This suggests that Y
2O
3 doping may decrease the fluidity of the glass phase, thereby preventing the formation of a continuous protective film, as corroborated by
Fig. 5i, j. As presented in
Fig. 5k-m, ZS20L5 exhibits a flatter and denser oxidized surface compared with other samples, with the glass phase primarily composed of B-Si-O-La system. The area uncovered by the glass phase is mainly ZrO
2 particles, and the borosilicate glass phase is uniformly filled between the ZrO
2 particles, which effectively blocks the diffusion of oxygen. This also explains why Y
2Si
2O
7 has the highest oxygen adsorption energy compared to La
2Si
2O
7, the oxidation resistance of ZS20Y5 is inferior to that of ZS20L5. Oxidation cross-sections in Fig. S7 demonstrate that incorporating rare earth oxides decreases the oxidation layer depth compared to ZS20. Among these samples, ZS20L5 exhibits the densest oxide layer and the shallowest oxidation depth. In summary, the addition of La
2O
3 results in the optimal comprehensive effect of the glass phase, which is most conducive to improving ZrB
2-SiC oxidation resistance.