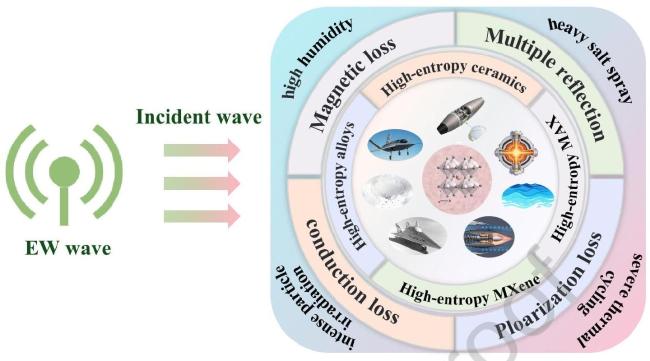
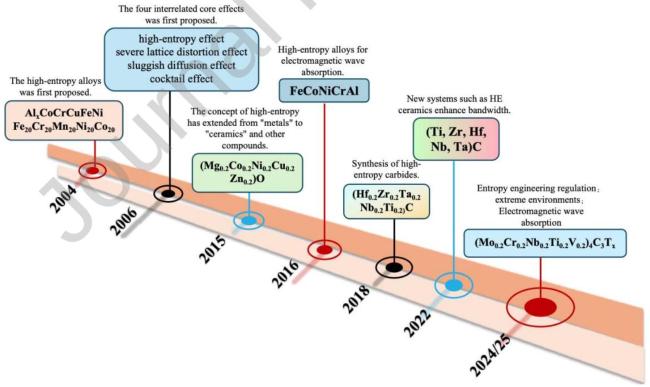
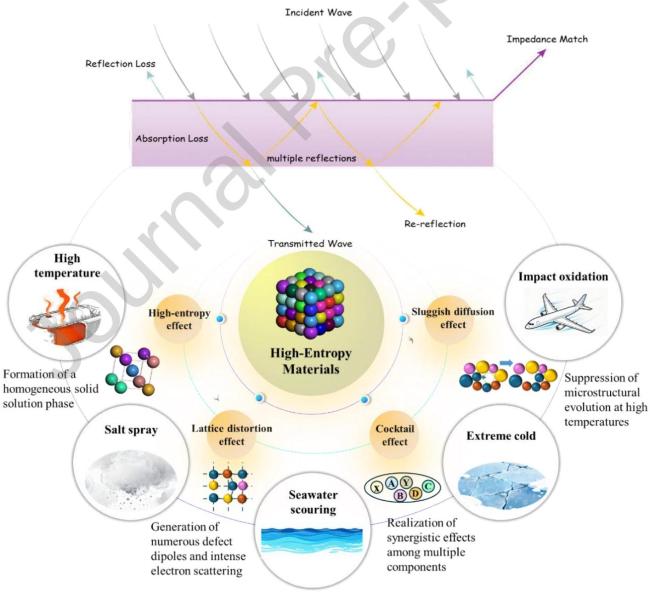
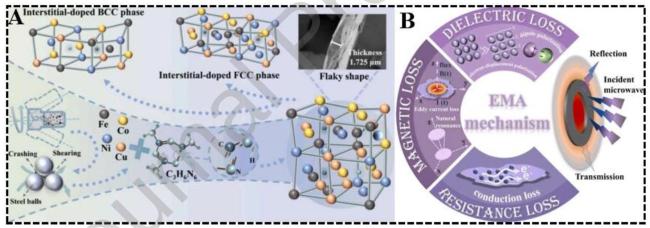
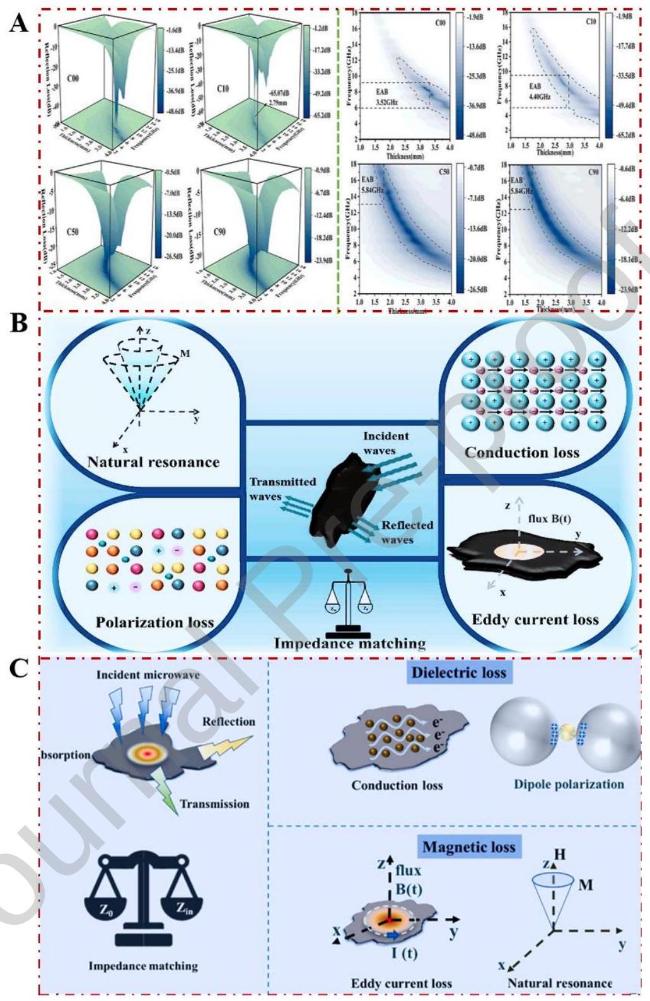
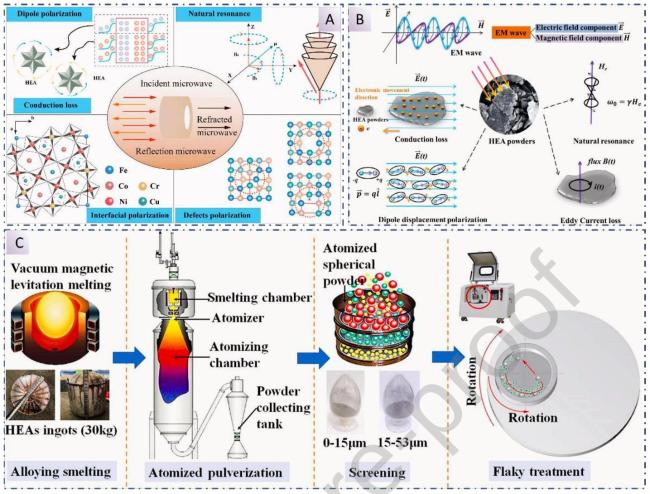
1. Introduction
Fig. 1 Application of High-Entropy Materials in Electromagnetic Wave Absorption under Extreme Environments. |
2 Core effects of HEMs and their synergistic mechanisms in electromagnetic wave (EW) absorption
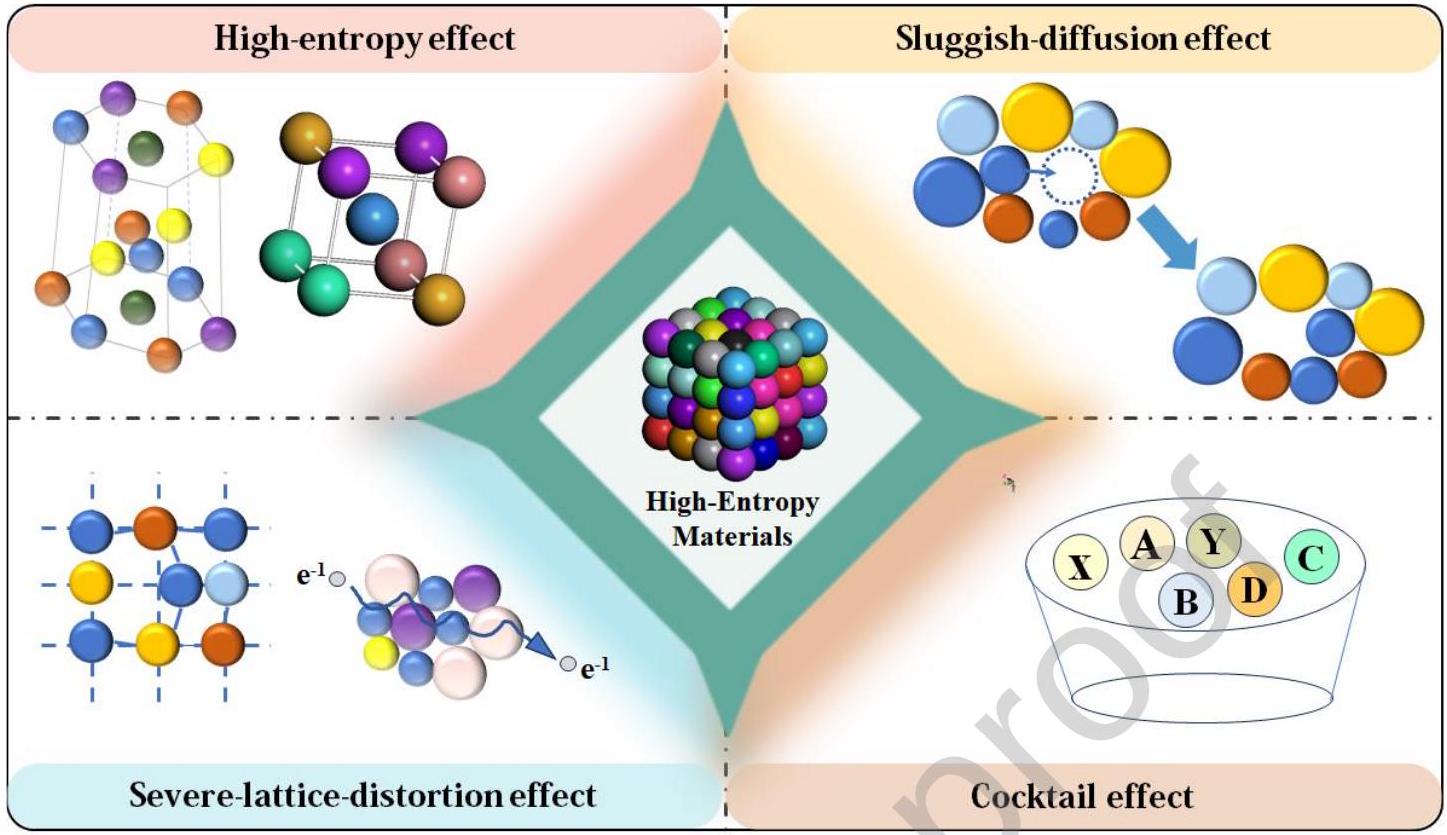
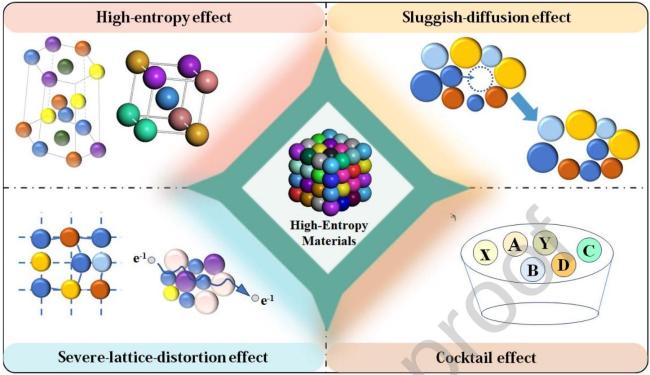
2.1 Typical characteristics and the four core effects of HEMs
Fig. 2 The four core effects in high-entropy materials |
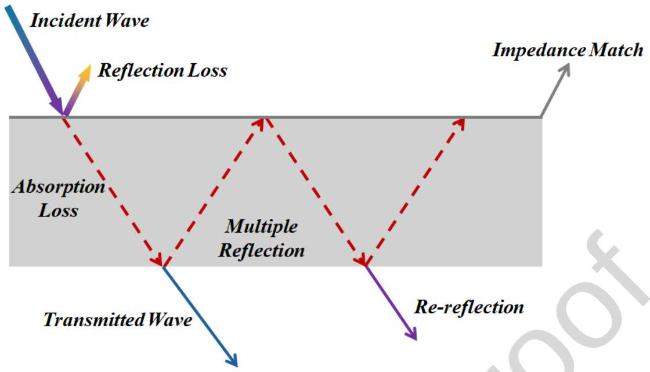
2.2 Fundamental mechanisms of electromagnetic wave absorption
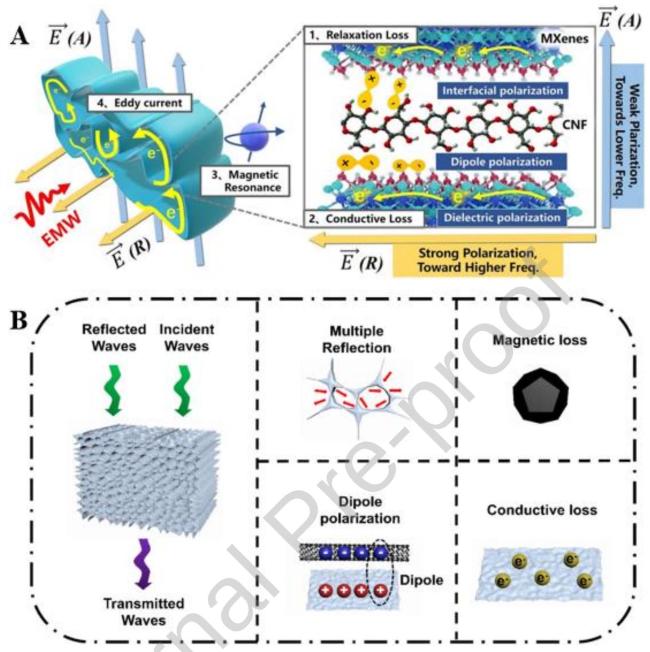
Fig. 3 Schematic diagram of the loss of incident electromagnetic wave through absorbing material. |
Fig. 4 Advances in High-Entropy Materials for Electromagnetic Wave Absorption |
2.3 Synergistic bridging: how high-entropy characteristics govern EW absorption
Fig. 5 The unique effects of high-entropy materials enable exceptional absorption performance under extreme conditions. |
3 High-entropy alloys
3.1 Compositional design and optimization
3.2 Synergistic design of surface engineering and composite architectures
Fig.6 Schematic illustrations of the preparation of carbonitrided FeCoNiCu high-entropy alloy sheets and their electromagnetic absorption mechanism. Reproduced with permission from Ref. [94], © Elsevier 2024. |
Fig. 7 (A)Three-dimensional (3D) RL, plots and 2D projections for different samples at various thicknesses. Reproduced with permission from Ref. [95], © Elsevier 2024. (B)Carburized FeCoNiMn HEAs. Reproduced with permission from Ref. [95], © Elsevier 2024. (C) Phosphated FeCoNiMn HEAs. Reproduced with permission from Ref. [89], © Elsevier 2024. |
3.3 Process innovation and performance optimization
Fig. 8 (A) FeCoNi HEA electromagnetic loss mechanism diagram. Reproduced with permission from Ref. [97], (C) Elsevier 2025. (B)Schematic illustration of the interaction between alloy powders and electromagnetic wave. Reproduced with permission from Ref. [98], © Elsevier 2019. (C)Schematic of the preparation process of HEA flaky powder. Reproduced with permission from Ref. [99], © Elsevier 2025. |
4 High-entropy ceramics
4.1 High-entropy oxides
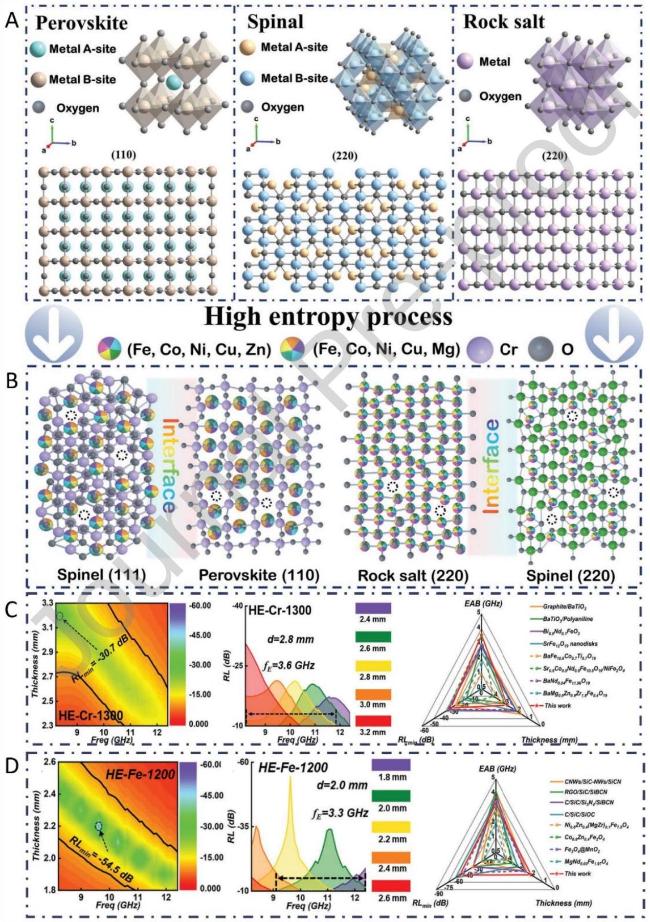
Fig. 9 (A) Crystal structure of perovskite, spinel and rock-salt phases with cubic structure (Fd-3m). (B) Diagram of defect, hetero-interface and lattice distortion in dual-phase HE oxides.(C) RL values of HE-Cr-1300 at X-band with various thicknesses. (D) RL values of HE-Fe-1200 at X-band with various thicknesses. Reproduced with permission from Ref. [110], © Wiley-VCH GmbH 2022. |
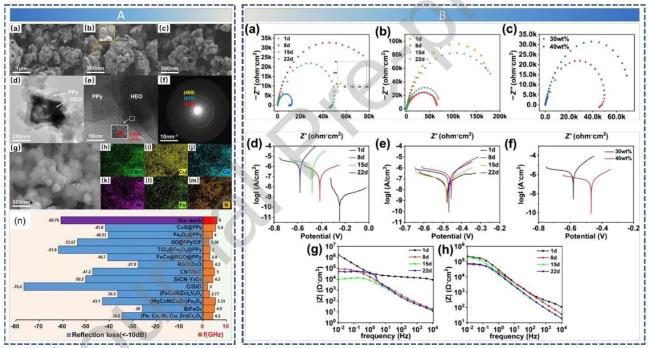
Fig. 10 (A) Microstructure Characterization and Performance Benchmarking of the HEO-based Absorber. (B) Nyquist, Tafel polarization, and Bode plots of materials with different mass ratios after various days of corrosion. Reproduced with permission from Ref. [111] , © Elsevier 2025. |
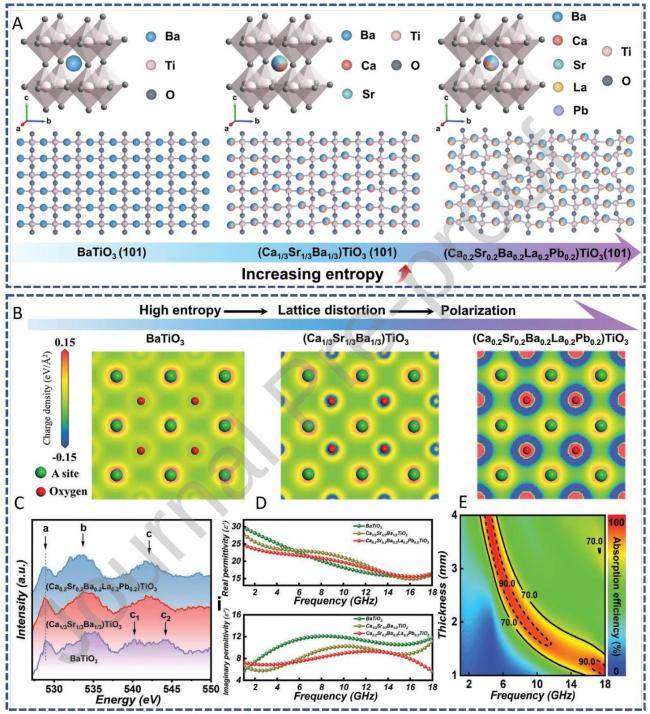
Fig. 11 summarizes the theoretical and experimental characterization, simulated charge distribution, O K-edge ELNES, permittivity, and microwave absorption performance of high-entropy titanate perovskites. Reproduced with permission from Ref. [112], © Wiley-VCH GmbH 2023. |
4.2 High-entropy carbides
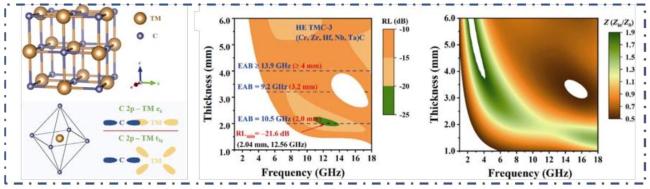
Fig. 12 Microstructure and Electromagnetic Properties of High-Entropy TMCs. Reproduced with permission from Ref. [120]. |
4.3 High-entropy borides
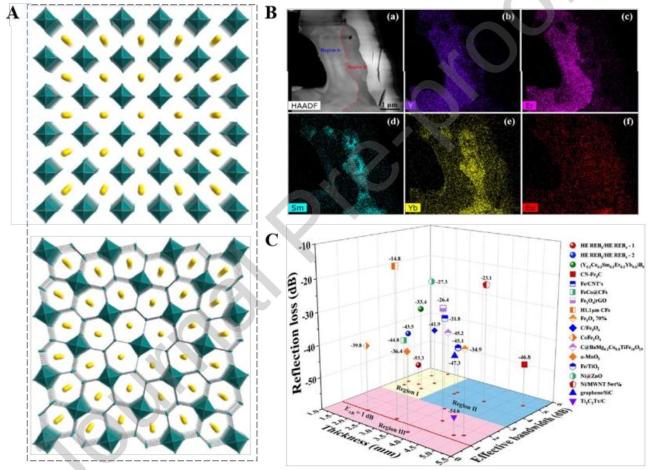
Fig. 13 Crystal structure of (A)REB 6 and REB 4 . (B)HAADF-STEM image of the high-entropy REB powder along with corresponding elemental distribution maps. (C)Comparison of electromagnetic wave absorption performance. Reproduced with permission from Ref. [128], © Elsevier 2021. |
5 High-entropy MAX/MXene
5.1 Preparation and microwave absorption properties of high-entropy MAX
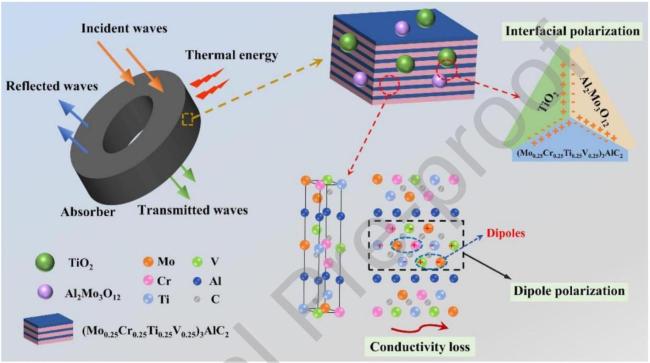
Fig. 14 Schematic illustration of the Ey wave absorption behaviors and mechanisms for MCTV- 800 powders. Reproduced with permission from Ref. [44], © Elsevier 2023. |
5.2 Preparation and microwave absorption properties of high-entropy MXenes
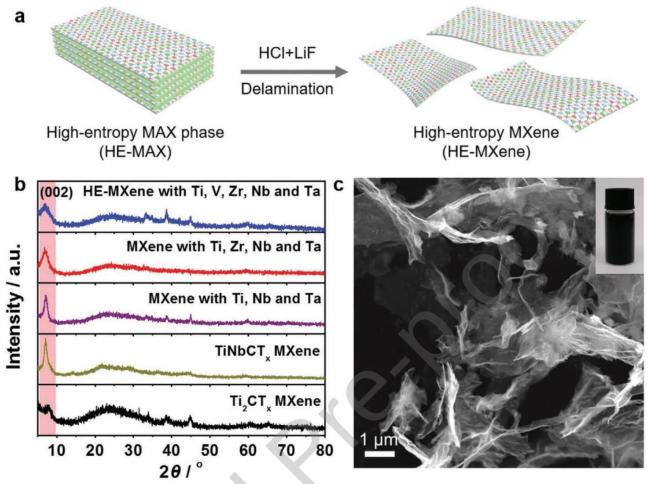
Fig. 15 Morphology and structure characterization of high-entropy MXene. Reproduced with permission from Ref. [129], © Wiley-VCH GmbH 2021. |
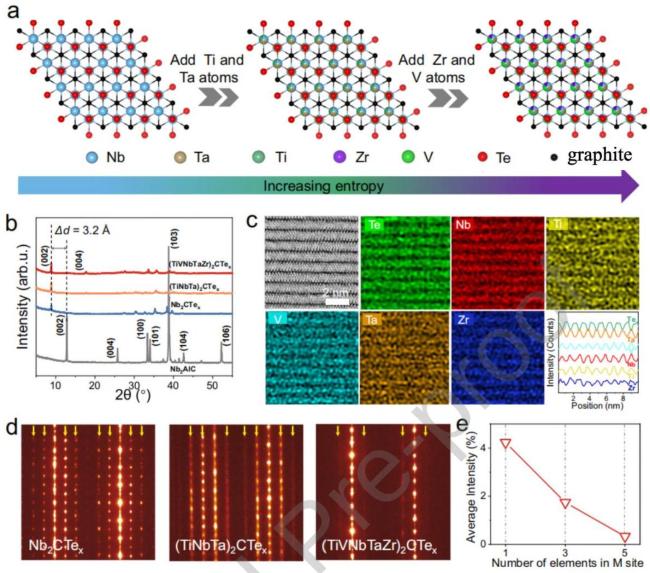
Fig. 16 illustrates the lattice evolution, XRD patterns, atomic-resolution elemental mappings, SAED patterns, and the effect of M-site element number on the diffraction intensity of high-entropy MXenes. Reproduced with permission from Ref. [130]. |
Table 1. Comparison of Electromagnetic Wave Absorption Performance for High-Entropy Materials under Extreme Environments |
| No. | Material System | RLmin (dB) | EAB (GHz) | Thickness (mm) | Structure/Treatment | Sample Pretreatment | Reference |
|---|---|---|---|---|---|---|---|
| 1 | FeCoNiCuTi0.2 | -47.8 | 4.76 | 2.16 | Flaky | [87] | |
| 2 | FeCoNiMn@P | -62.4 | 4.1 | 2.1 | Phosphorization | [89] | |
| 3 | FeCoNiCu(C,N) | -61.8 | 3.82 | 2.38 | CarbonNitrogen Co-diffusion | [94] | |
| 4 | FeCoNiMnCx(C10) | -65.07 | 5.84 | 2.79 | Carburization | [95] | |
| 5 | FeCoNiCrMn/PLA | -24.58 | 2.51 | 4.5 | 3D Printing | Seawater + Oil pollution | [96] |
| 6 | FeCoNi1.5 CuCr | -46.3 | 4.1 | 2.0 | Magnetic field treatment (6 T) | 6 T pulsed magnetic field | [97] |
| 7 | FeCoNiAlCr0.9 | -47.55 | 6.6 | 1.9 | Ball Milling + Annealing | Annealing at | [98] |
| 8 | FeCro. 5 NiCu0.5 | -26.1 | 3.6 | 1.2 | Gas Atomization + Ball Milling | Marine salt | [99] |
| 9 | FeCoCrMn | -59.6 | 6.86 | 1.32 | Multi-element (C, N, O) Penetration | [100] | |
| 10 | TiVNbMoC3Tx/Fe3O4 | -57.59 | 4.72 | 1.46 | 0D/2D hybrid, electrostatic self-assembly | Oxidation | [132] |
| 11 | (Mo0.25Cr0.25Ti0.25V0.25)3AlC2 | -45.80 | 3.6 | 1.7 | High-entropy MAX phase, solid-state synthesis | Oxidation ( , for 1 h ) | [44] |
Table 2. summarizes several major categories of high-entropy materials (alloys, oxides, ceramics, carbides, borides, MAX/MXene), outlining their core advantages and main challenges. High-entropy materials generally exhibit good structural stability, strong tunability of properties, and high environmental adaptability. However, they also face issues such as high density, brittleness, difficulties in synthesis and processing, insufficient stability, or being in the early stages of research. Future development requires further breakthroughs in process optimization and long-term reliability. Table 2.High-Entropy Materials for Electromagnetic Wave Absorption in Extreme Environments:A Comparative Summary of Strengths and Challenges |
| Material Category | Main Advantages | Key Limitations / Challenges |
|---|---|---|
| High-entropy alloys | 1. Excellent structural stability: Resulting from the combined effects of the high-entropy effect and sluggish diffusion effect. 2. Strong tunability of properties: The “cocktail effect” facilitates the achievement of broadband and tunable wave-absorbing properties through compositional design. 3. Flexible processing; | 1. Long-term service reliability requires further verification; 2. Relatively high density. |
| High-entropy oxides | 1. Exceptional thermal stability and oxidation resistance; 2. Excellent corrosion resistance:The ceramic nature inherently provides superior corrosion resistance. | 1. Typically, low electrical conductivity; 2. High brittleness and poor toughness. |
| High-entropy ceramics | 1.Extreme environmental adaptability; 2. Broadband and efficient absorption:Lattice distortion and defect states significantly enhance polarization loss. | 1. Extremely high synthesis temperature; 2. Difficulties in processing and shaping. |
| High-entropy Borides | 1. Outstanding comprehensive performance:Combines ultra-high melting point,high hardness,good thermal conductivity,and oxidation resistance; 2. Easy tuning of impedance matching. | 1.Research is still in its early stages; 2. Immature composite processing technology. |
| High-entropy MAX/MXene | 1. Unique structure-function characteristics:The layered structure can be easily exfoliated into MXene; 2. Good environmental adaptability. | 1. Stability issues:Multilayer MXene is prone to oxidation in humid or high-temperature environments,and long-term stability remains an application bottleneck. 2. Etching process is complex and hazardous. |