| Isotherm | Isotherm Equations | Temp. (K) | Equation | R2 |
|---|---|---|---|---|
| Langmuir | | 298 | y=0.9763x+0.0023 | 1 |
| 308 | y=0.9761x+0.0253 | 1 | ||
| 318 | y=0.9680x+0.0200 | 1 | ||
| 328 | y=0.9675x+0.0651 | 0.9999 | ||
| Freundlich | | 298 | y=0.0237x-0.0023 | 0.9963 |
| 308 | y=0.0239x-0.0253 | 0.9430 | ||
| 318 | y=0.0320x-0.0200 | 0.9588 | ||
| 328 | y=0.0325x-0.0651 | 0.9188 | ||
| Temkin | | 298 | y=4.4x+98.15 | 0.9903 |
| 308 | y=4.09x+92.66 | 0.9552 | ||
| 318 | y=5.17x+92.78 | 0.9676 | ||
| 328 | y=5.01x+84.89 | 0.8871 | ||
| Flory-Huggins | | 298 | y=6.8517x+9.3482 | 0.8874 |
| 308 | y=12.501x+12.123 | 0.9779 | ||
| 318 | y=11.166x+10.501 | 0.9769 | ||
| 328 | y=10.479x+8.9185 | 0.8040 | ||
| El-Awady | | 298 | y=0.1502x+1.2984 | 0.9413 |
| 308 | y=0.1003x+0.9398 | 0.9753 | ||
| 318 | y=0.1168x+0.9144 | 0.9790 | ||
| 328 | y=0.1071x+0.7213 | 0.8473 | ||
| Frumkin | | 298 | y=-43.807x+46.103 | 0.9771 |
| 308 | y=-48.207x+47.232 | 0.9480 | ||
| 318 | y=-37.939x+37.595 | 0.9610 | ||
| 328 | y=-35.748x+33.068 | 0.8531 |
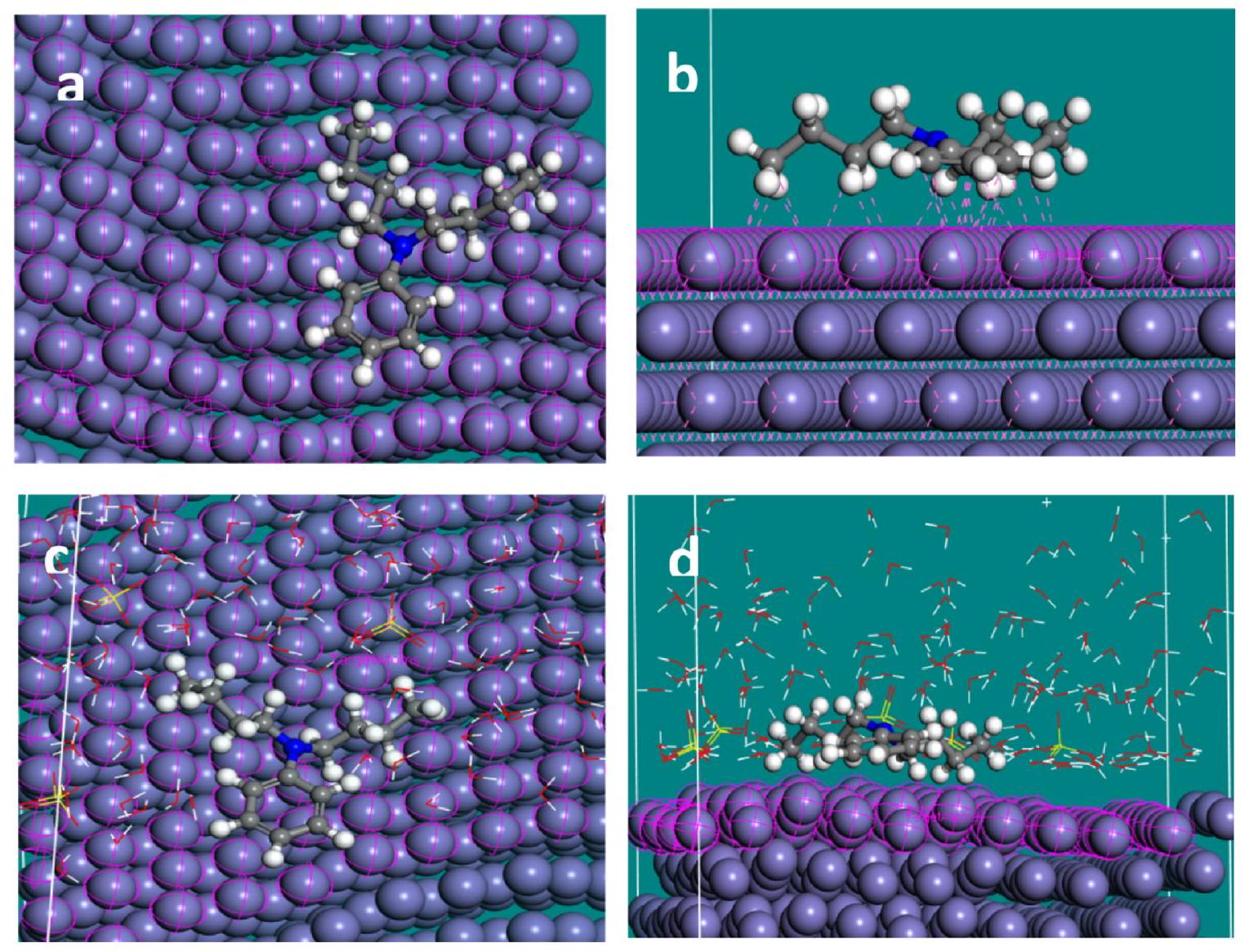 Fig. 16. MD simulation captures in (a) top view of adsorbed NNDBA molecule on mild steel surface (b) front view of interactions between NNDBA and mild steel (c) top view of adsorbed NNDBA molecule on mild steel in presence of H2O and H2SO4 (d) top view of adsorbed NNDBA molecule on mild steel in presence of H2O and H2SO4.
Fig. 16. MD simulation captures in (a) top view of adsorbed NNDBA molecule on mild steel surface (b) front view of interactions between NNDBA and mild steel (c) top view of adsorbed NNDBA molecule on mild steel in presence of H2O and H2SO4 (d) top view of adsorbed NNDBA molecule on mild steel in presence of H2O and H2SO4.